In 1957, the residents of Tulsa, Oklahoma buried an enormous time capsule that included a Plymouth Belvedere and containers of gasoline. The residents, at that time, thought that the folks in 2007 might not recognize the common mode of transportation of their time and would certainly not have the oil or gasoline to make it run.
While 60 years later, we are still driving automobiles powered by gas and oil, there are some modern-day products that are in danger of extinction due to the lack of metals and elements used in their manufacturing process and operations.
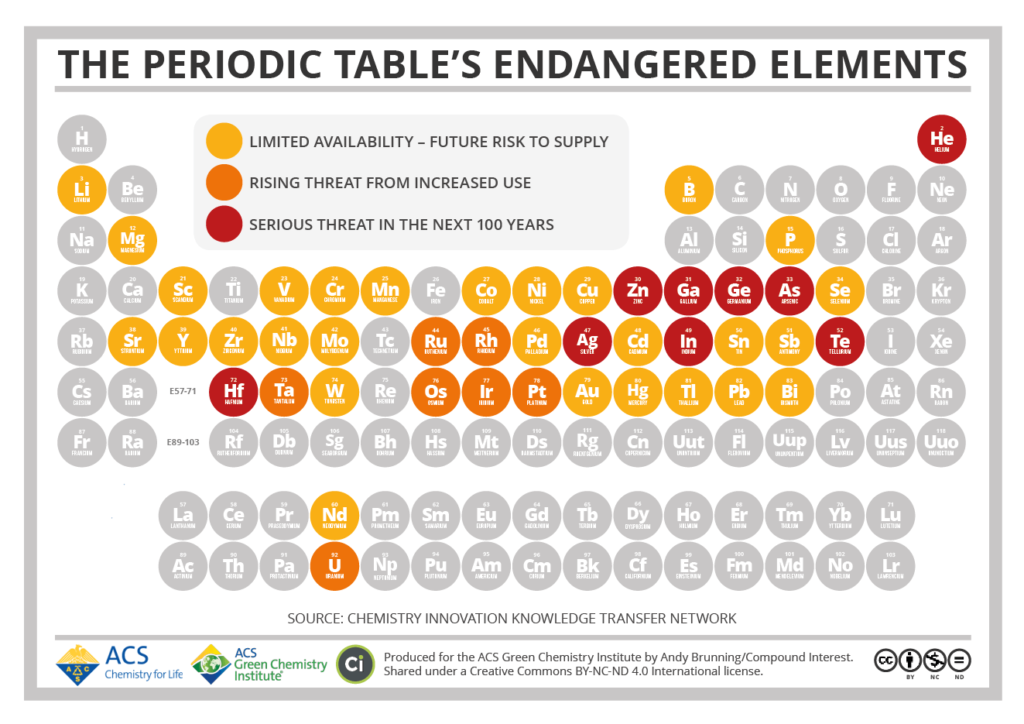
Highlighted on the above Periodic Table are endangered elements that currently have limited availability, are considered to be a rising threat from increased use, and are in serious threat of depletion over the next 100 years. A few of the top elements and their use are discussed in this brief article.
Lithium
Lithium is a soft gray clay-like metal that can be cut with a butter knife. For decades, lithium has been used in everything from glass and ceramics to grease and polymers. It is even used in medicine to treat bipolar disorder.
However, a growing use of this highly conductive metal is in the manufacturing of batteries. There are many types of batteries made of Nickel Cadmium (NiCd), Nickel-Metal Hydride (NiMH), Lead Acid, Lithium Ion, and Lithium Polymer. But, when it comes to battery performance, lithium rises to the top of the list because of its ability to be drained and then recharged over a period of hundreds of cycles. Lithium batteries are the choice for the new electric automobile industry, as well as, the solar industry.
And while batteries make up 40 percent of the current lithium use, that number is expected to grow to over 65 percent in the next 10 years. According to Credit Suisse, demand could outstrip supply by 25 percent in the next three years.
Rare-Earth Elements
Rare-Earth is a category of elements, some of which are not identified in the periodic table, that are essential for the medical/pharma industry, as well as, manufacturing, industrial, and especially electronics.
The issue with rare-earth elements is not their lack of abundance, but rather, isolating them. They tend to occur together, and separating them from each other is made difficult by the fact that they all have very similar chemical properties.
As with many of the other elements on this list, the demand for rare earth elements is predicted to be fast outpacing the supply, and it’s a problem that’s yet to be solved.
As computers, automobiles, and other manufactured products come to the end of their useful life, many scientists feel that recycling is the answer to recapturing some of these quickly diminishing materials. However, coming up with economically viable methods for recovery of these often minuscule quantities of material is a huge challenge. Finding a substitute substance is also an option, but in most cases to date, the product quality suffers.
As we look back, the world became mechanized by the Industrial Revolution of the 18th and 19th centuries. The 20th century brought computerization to businesses and the miniaturization of electronics for the masses. However, those three centuries alone caused more destruction to the environment and consumed more natural resources than the previous 17 centuries. Now, as we near the end of the second decade of the 21st century, the question will be whether man can turn the tide of destruction by learning to conserve and perhaps recycle our precious natural resources before they disappear?
To learn more about the conservation of earth’s precious elements and the steps you can take to reduce, reuse and recycle – contact General Kinematics.